
When AI Truly Makes a Difference: 5 Real Breakthroughs in Medicine
Artificial intelligence is often described as “the future of medicine.”
The reality is more interesting: in many areas, AI is already operational, sometimes for years, and is improving diagnoses, therapies, and clinical workflows in tangible and measurable ways.
In this article, we explore five significant, well-documented cases where AI has represented a real turning point, with references to studies and concrete technical implementations.
1. Early Cancer Detection Through AI-Based Medical Imaging
Medical imaging is one of the areas where artificial intelligence has delivered some of the most immediate and concrete results. A research group led by the Mayo Clinic demonstrated that AI models can detect early signs of pancreatic cancer in standard abdominal CT scans up to three years before clinical diagnosis. The system was trained on tens of thousands of patient scans, learning to identify subtle patterns that are often invisible to the human eye.
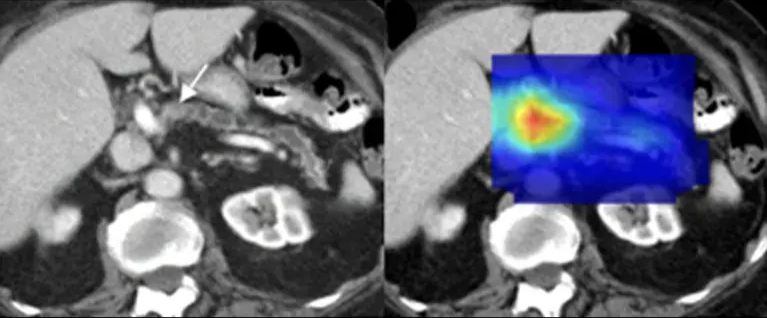
AI-assisted analysis of abdominal CT scans highlights microscopic pancreatic patterns, enabling cancer detection years before traditional diagnostic methods.
A complementary study published on SpringerLink shows that AI can also significantly improve diagnostic sensitivity in thoracic and neuro-radiology, for example by increasing the detection rate of lung nodules or lesions associated with multiple sclerosis.
Nerd corner
These systems rely on deep learning models specifically designed to “read” medical images. In practice, 2D and 3D convolutional neural networks (CNNs) are trained on large datasets of annotated CT scans to recognize complex morphological patterns at the pixel level, similar to how a radiologist works, but with much finer granularity. U-Net architectures are commonly used for semantic segmentation, allowing the model to automatically isolate potentially pathological regions. The output is not a simple yes/no classification, but heatmaps integrated into PACS systems that show where the model is focusing its attention. Performance is evaluated using screening-oriented metrics such as AUC, sensitivity, and false-negative rate, critical measures when the goal is early disease detection.
2. Predicting Alzheimer’s Disease Before Clinical Symptoms Appear
One of the most promising applications of AI in medicine is the early prediction of neurodegenerative diseases, particularly Alzheimer’s. Here, the value of AI lies less in formal diagnosis and more in its ability to identify weak signals years before cognitive decline becomes clinically evident.
Several studies conducted in the United States, including research from the University of California San Francisco (UCSF), show that machine learning models can estimate the risk of developing Alzheimer’s five to seven years before symptoms appear by integrating longitudinal clinical data, neuroimaging, and biomarkers.
In practice, AI is used to detect progression patterns that may be insignificant in isolation but, when combined, reveal a clear risk trajectory.
Nerd corner
The most effective approaches work on longitudinal data, information collected over time from the same patient. Models typically combine supervised machine learning algorithms (such as random forests and gradient boosting) for structured electronic health records with deep neural networks applied to brain MRI or PET scans. When the goal is to estimate when a patient might develop the disease, AI-enhanced survival analysis models are used to associate risk profiles with temporal windows. In more advanced research settings, linguistic and cognitive signals are also integrated via transformer models to detect early semantic changes.
3. Discovering New Drugs with Generative AI
In 2020, MIT made headlines with the discovery of Halicin, a novel antibiotic identified by an AI system that screened over 100 million known molecules in just a few days. Published in Cell, the study showed how generative models can uncover therapeutic candidates that traditional approaches would likely miss.
At the same time, biotech company Insilico Medicine used its proprietary platforms, PandaOmics and Chemistry42, to design and advance an anti-fibrotic drug into clinical trials in less than 30 months, compared to an industry average of five to six years.
Nerd corner
In drug discovery, AI is used to explore vast chemical spaces that are impractical to navigate manually. Generative models such as variational autoencoders (VAEs) and GANs learn the “grammar” of molecules and generate new candidate structures. These candidates are then evaluated using predictive models (QSAR) and molecular docking simulations. Optimization is often guided by reinforcement learning, balancing multiple objectives at once, efficacy, toxicity, stability, and synthesizability. The result is not a replacement for the lab, but a dramatic reduction in the number of experiments needed.
4. AI-Assisted Surgery: More Precision, Less Variability
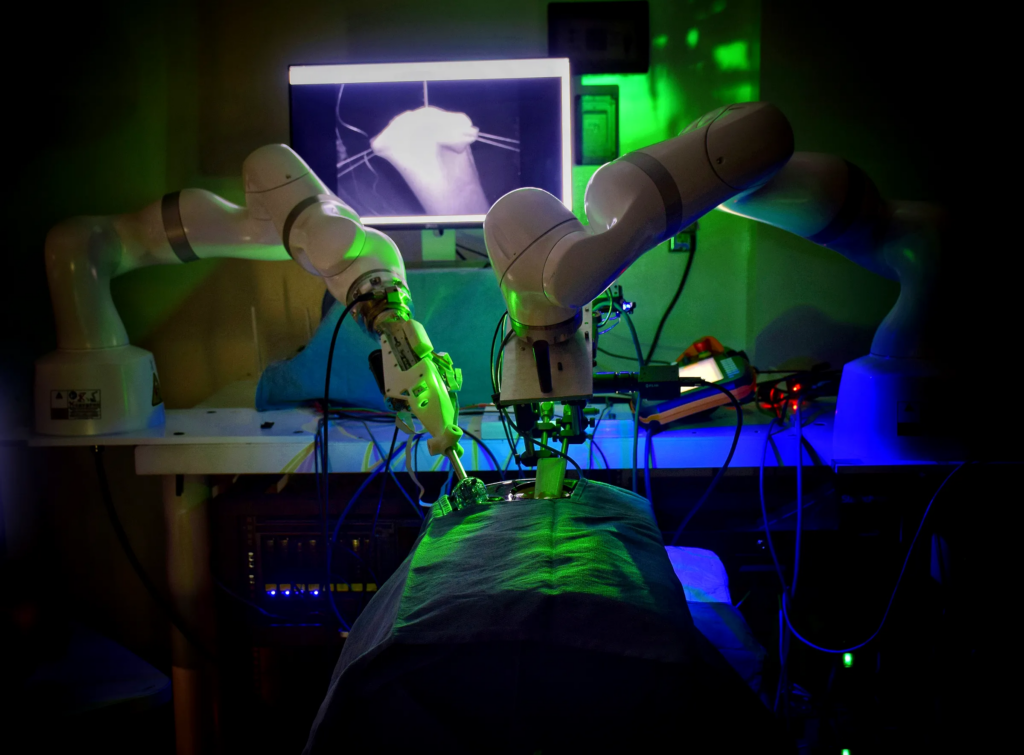
AI-assisted robotic systems are already transforming surgical practice. Platforms such as ROSA (used in neurosurgery and orthopedics) integrate pre-operative imaging, personalized planning, and real-time feedback to improve precision. Even more striking is the case of STAR, a robot capable of autonomously suturing soft tissue, outperforming human surgeons in controlled studies published in Science Translational Medicine.
The STAR surgical robot uses computer vision and AI algorithms to perform autonomous soft-tissue suturing, adapting in real time to tissue movement.
Nerd corner
In surgical robotics, AI must operate in a constantly changing, deformable environment. Systems combine 3D computer vision, tissue tracking, and motion-planning models inspired by human surgical trajectories. Training relies on datasets of real surgical procedures, from which the system learns gestures, constraints, and variability. During surgery, control algorithms continuously adjust trajectories based on visual, force, and in some cases, fluorescence imaging feedback, enabling adaptive and reproducible suturing.
5. Personalized Medicine and Complex Cases: AI-Based Decision Support
In oncology and rare diseases, AI is reshaping therapeutic decision-making. The ACMA (AI Case Matching Algorithm), presented at ASCO, analyzes thousands of clinical records, genetic profiles, and treatment outcomes to suggest personalized therapy options with greater accuracy than traditional approaches.
A well-known example shared by physician and digital health expert Bertalan Meskó describes a patient with a refractory form of Castleman disease, where an AI-assisted approach helped identify an off-label drug that ultimately led to remission.
Nerd corner
In clinical decision support, the goal is not just prediction, but relationship modeling. Multimodal systems and graph-based approaches represent diagnoses, genes, drugs, treatments, and outcomes as interconnected nodes. Techniques such as graph embeddings and graph neural networks help identify patterns and similarities across complex cases, enabling physicians to compare a patient against thousands of real-world examples. Explainability is essential: every recommendation must be traceable and justifiable in a real clinical context.
_What We Learn from the Medical Approach to AI
These cases show that AI in medicine is no longer experimental, it is already a high-impact enabling technology.
What is often underestimated, however, is that training data quality and provenance make all the difference between theoretically promising AI and systems that truly work in clinical practice.
In healthcare, AI models do not learn in isolation. They are trained on years of human clinical expertise: diagnostic images annotated by specialists, structured medical records, and validated surgical procedures. The STAR robot, for example, does not “learn on its own”, its capabilities derive from analyzing thousands of sutures performed by human surgeons, transformed into data, trajectories, and reproducible patterns. The same applies to imaging systems trained on radiologist-annotated datasets or predictive models that encode historical clinical knowledge.
That is why, in medicine, AI does not replace human expertise, it absorbs it, amplifies it, and makes it scalable. Without reliable, representative, and well-curated data, even the most advanced algorithm remains ineffective.
At Fyonda, we believe in explainable, concrete, and responsible AI, built on high-quality data and designed to support real decisions. The challenge is not only technological, but cultural: integrating AI in an ethical, safe, and sustainable way, while respecting the human expertise from which it originates.




